Nuclear Chemistry Review
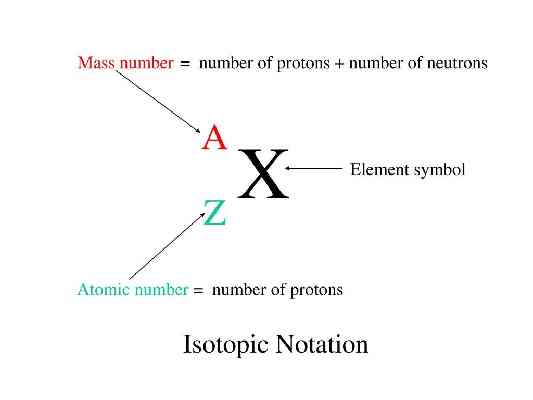
{"name":"Nuclear Chemistry Review", "url":"https://www.quiz-maker.com/QWH0IS7BI","txt":"radioactivity - emission of ionizing (dislodge e-) radiation by some of the heavier elements TO REACH STABILITY; can be particulate or non-particulate in nature, alpha particle - charge (+) beta particle - charge (-) gamma ray - no charge with these, which are influenced by electric\/magnetic field?, (A.) lowest penetration but highest biological distraction; (B.) highest penetration but lowest biological distraction","img":"https://www.quiz-maker.com/3012/images/ogquiz.png"}